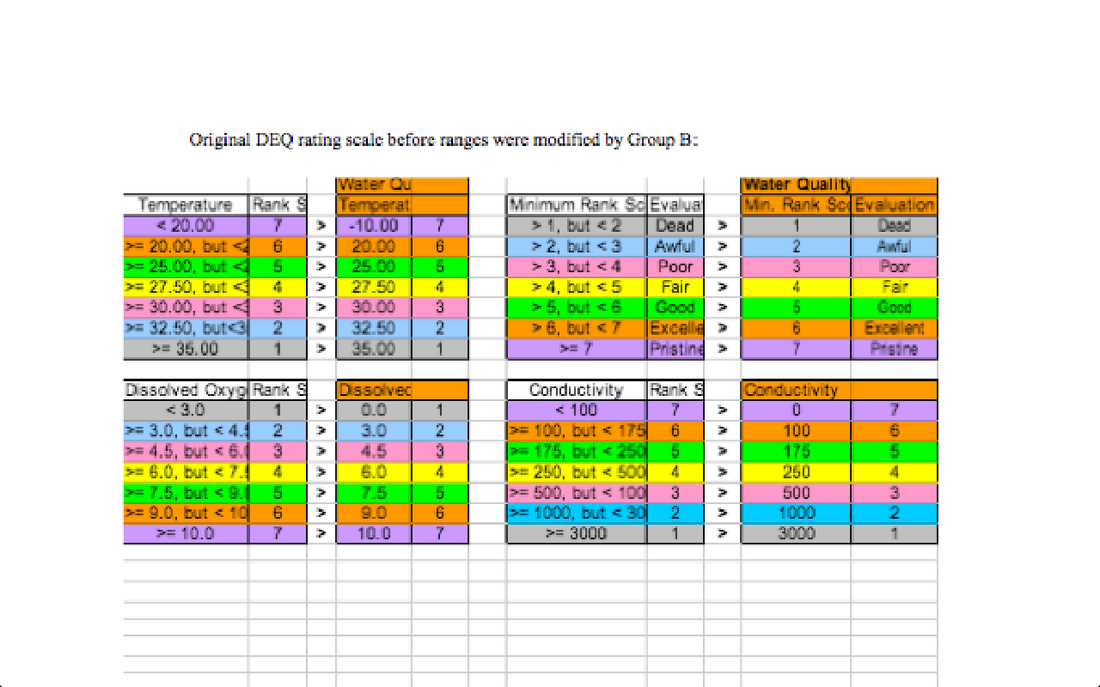
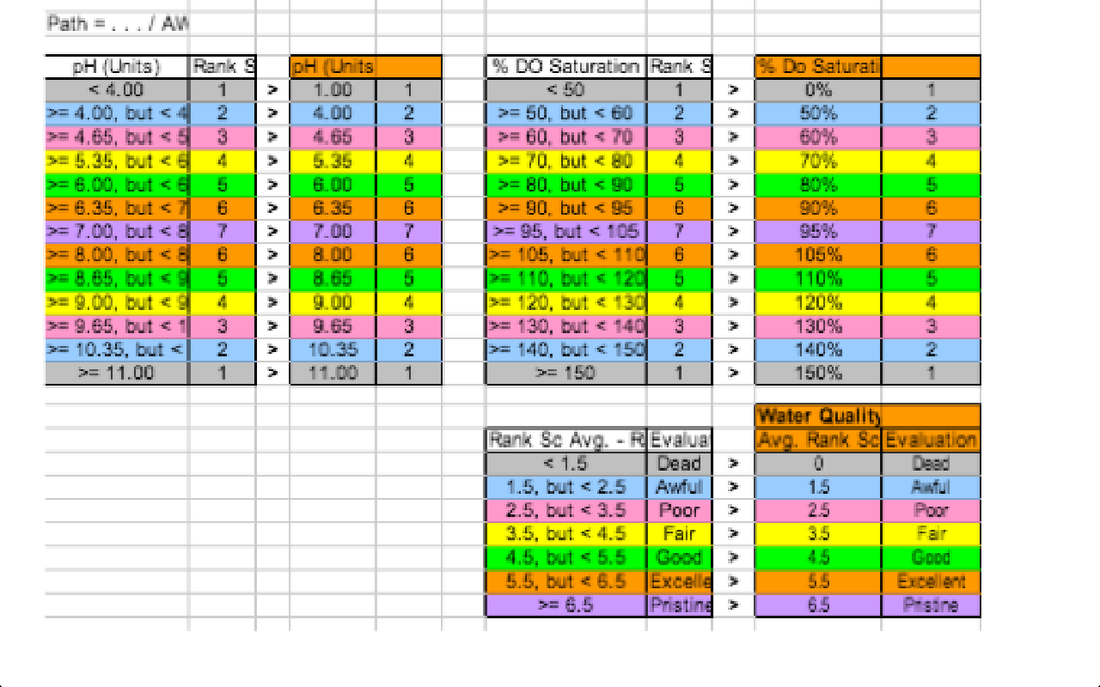
The Importance of each parameter to the health of a freshwater stream-
DO
Dissolved oxygen (DO) is a measure of how much oxygen is dissolved in the water. The oxygen dissolved in lakes, rivers, and oceans is crucial for aerobic respiration in benthic organisms as well as fish and other large aquatic creatures. As the amount of dissolved oxygen drops, the water quality worsens and the organisms begin to die off. Rapidly moving water, such as in a mountain stream or large river, tends to contain a lot of dissolved oxygen due to aeration, whereas stagnant water contains less. Because there is less kinetic energy in cold water, it can hold more oxygen than warm water. In the water bacteria can consume oxygen as organic matter decays. Thus, excess organic material in lakes and rivers can cause eutrophic conditions, which is an oxygen-deficient situation that can cause aquatic organisms to perish. A dissolved oxygen level that is too high or too low can harm aquatic life and affect water quality. A cold mountain stream with a rock bottom will hold more oxygen than a warm slow-moving creek. (USGS)
pH
The pH of river water is the measure of how acidic or basic the water is on a scale of 0-14. Specifically, it is a measure of hydrogen ion concentration. The optimum pH for river water is around 7.4. Water's acidity can be increased by acid rain (pH below 7) but is kept in check by the buffering limestone. Acidic water also increases the leaching of ions. This results in the increase of some toxic metals (lead, mercury, cadmium) that are harmful to the nervous system of fish. Extremes in pH can make a river inhospitable to life. Low pH is especially harmful to immature fish and insects. (NASA)
Temperature
Temperature exerts a major influence on biological activity and growth. Temperature governs the kinds of organisms that can live in rivers and lakes. Fish, insects, zooplankton, phytoplankton, and other aquatic species all have a preferred temperature range. As temperatures get too far above or below a preferred range, the number of individuals of the species decreases until they diminish completely. Temperature is also important because of its influence on water chemistry. The rate of chemical reactions generally increases at higher temperature. Warm water holds less dissolved oxygen than cool water, and may not contain enough dissolved oxygen for the survival of different species of aquatic life. Some compounds are also more toxic to aquatic life at higher temperatures. Based on our evidence, both warm water and cold water each have their own separate but beneficial influences on the aquatic environment. (USGS)
BOD
Biochemical oxygen demand, or BOD, measures the amount of oxygen consumed by microorganisms in decomposing organic matter in stream water. BOD also measures the chemical oxidation of inorganic matter (i.e., the extraction of oxygen from water via chemical reaction). The rate of oxygen consumption in a stream is affected by a number of variables: temperature, pH, the presence of certain kinds of microorganisms, and the type of organic and inorganic material in the water. BOD directly affects the amount of dissolved oxygen in rivers and streams. The greater the BOD, the more rapidly oxygen is depleted in the stream. This means less oxygen is available to higher forms of aquatic life. The consequences of high BOD are the same as those for low dissolved oxygen: aquatic organisms become stressed, suffocate, and die. Reasons for increased BOD include leaves and woody debris; dead plants and animals; animal manure; effluents from pulp and paper mills, wastewater treatment plants, feedlots, and food-processing plants; failing septic systems; and urban stormwater runoff. (EPA)
Nitrate
Nitrates are a form of nitrogen, which is found in several different forms in terrestrial and aquatic ecosystems. These forms of nitrogen include ammonia (NH3), nitrates (NO3), and nitrites (NO2). Nitrates are essential plant nutrients, but in excess amounts they can cause significant water quality problems. Based on the quantity of the nitrate in the river, we are given a good indication to whether or not the river is healthy. Together with phosphorus, nitrates in excessive amounts can accelerate eutrophication, causing dramatic increases in algal growth and subsequent decomposition. Excess nitrates can cause hypoxia (low levels of dissolved oxygen) and can become toxic to warm-blooded animals at higher concentrations (10 mg/L) or higher) under certain conditions. The natural level of nitrates in surface water is typically low (less than 1 mg/L); in the effluent of wastewater treatment plants, it can range up to 30 mg/L. Sources of nitrates include wastewater treatment plants, runoff from fertilized lawns and cropland, failing on-site septic systems, runoff from animal manure storage areas, and industrial discharges that contain corrosion inhibitors. (EPA)
Phosphate
Phosphorus is naturally present in water, primarily as inorganic and organic phosphates. Phosphates can enter aquatic environments in several ways: from the natural weathering of minerals in the drainage basin, from biological decomposition, or as runoff from human activity in urban and agricultural areas. Inorganic phosphates are usually present in the environment in low concentrations, which limits plant growth. High inorganic phosphate levels come from man-made sources such as septic systems, fertilizer runoff and improperly treated waste-water. These phosphates enter the water as the result of surface runoff and bank erosion. Inorganic phosphates are measured as “reactive” phosphates, and we measured this form in our testing. An increase in nitrate levels is often linked to an increase in phosphate levels. Phosphates can also result in eutrophication. In the streams that we analyzed, the optimum phosphate level would be no more than 0.1mg/L. (Lehigh. edu)
Ammonia
The formula for ammonia, NH3, means it consists of one atom of nitrogen and three atoms of hydrogen. Like nitrates, ammonia may speed the process of eutrophication in waterways. Ammonia is toxic to fish and aquatic organisms, even in very low concentrations. When ammonia is present in water at high enough levels, it is difficult for aquatic organisms to sufficiently excrete the toxins, leading to toxic buildup in internal tissues and blood, and potentially death. High ammonia also reduces hatching and growth rates of fish. It also affects structural development and causes changes in tissues of gills, liver, and kidneys..(http://www.water-research.net)
Environmental factors, such as pH and temperature, can affect ammonia toxicity to aquatic animals. The levels of ammonia in a stream help one to draw a conclusion of the health of a steam. When levels reach 0.06 mg/L, fish can suffer gill damage. When levels reach 0.2 mg/L, sensitive fish like trout and salmon begin to die. Ammonia levels greater than approximately 0.1 mg/L usually indicate polluted waters. The danger ammonia poses for fish depends on the synergistic effect of the water’s temperature and pH, along with the dissolved oxygen and carbon dioxide levels. High ammonia is much more toxic to fish and aquatic life when water contains very little dissolved oxygen and carbon dioxide and is warm with a low pH.(h2ou)
Conductivity
Conductivity is a measure of the ability of water to pass an electrical current. The basic unit of measurement of conductivity is the mho or siemen. Conductivity in water is increased by the presence of inorganic dissolved ions such as chloride, nitrate, sulfate, phosphate and anions as well as or sodium, magnesium, calcium, iron, and aluminum and hydrogen cations (to a lesser extent). Organic compounds like oil, phenol, alcohol, and sugar do not conduct electrical current very well and therefore have a low conductivity when in water. Conductivity is also affected by temperature: the warmer the water, the higher the conductivity. Conductivity in streams and rivers is affected primarily by the geology of the area through which the water flows. Streams that run through areas with granite bedrock tend to have lower conductivity. This is because granite is composed of more inert materials that do not ionize (dissolve into ionic components) when washed into the water. On the other hand, streams that run through areas with clay soils tend to have higher conductivity because of the presence of materials that ionize when washed into the water. Groundwater inflows can have the same effects depending on the bedrock they flow through. (EPA)
Turbidity
Turbidity is a measure of water clarity (how much the material suspended in water decreases the passage of light through the water). Suspended materials include soil particles (clay, silt, and sand), algae, plankton, microbes, and other substances. These materials are typically in the size range of 0.004 mm (clay) to 1.0 mm (sand). Higher turbidity increases water temperatures because suspended particles absorb more heat. This, in turn, reduces the concentration of dissolved oxygen (DO) because warm water holds less DO than cold. Higher turbidity also reduces the amount of light penetrating the water, which reduces photosynthesis and the production of DO. Suspended materials can clog fish gills, reducing resistance to disease in fish, lowering growth rates, and affecting egg and larval development. As the particles settle, they can blanket the stream bottom, especially in slower waters, and smother fish eggs and benthic macroinvertebrates. Sources of turbidity include:
- Soil erosion
- Waste discharge
- Urban runoff
- Eroding stream banks
- Large numbers of bottom feeders (such as carp), which stir up bottom sediments
- Excessive algal growth. (EPA)
E. coli
E. coli is a naturally occurring bacteria that lives in the lower intestines of warm-blooded mammals. E. coli belongs to a group of bacteria, some of which are harmful, known as fecal coliform. E. coli is necessary for the digestion of food but its presence in rivers indicates fecal contamination. Though, E. coli does not necessarily harm the microorganisms in an aquatic environment, an excess amount of E. coli in a human will negatively affect one’s health. Common sources of E.coli include overflowing septic fields and sewage systems, and fecal matter from wildlife and pets. The Environmental Protection Agency had designated a federal standard that no single samples shall exceed 235 colonies/100 mL. There should be no E. coli in drinkable water. (CRK)
Intolerant benthic organisms
Benthic macroinvertebrates are routinely used as biological indicators of water quality and overall stream health because they vary significantly in their tolerance of water pollution. For example, mayflies, stoneflies and most types of caddis flies require relatively cool, clear, well-oxygenated water and are sensitive to environmental changes brought on by water degradation. Generally, these intolerant organisms are said to be members of Group I. Moderately tolerant or somewhat sensitive dragonfly nymphs and crayfish are members of Group II whereas leeches, aquatic worms and most true flies (i.e. black flies and midges) which can tolerate warmer, more turbid conditions comprise Group III. Considered as a whole, the ratio of tolerant to intolerant organisms can indicate the overall health of the waterway. Unlike fish, macroinvertebrates can’t escape sudden changes in the aquatic environment by swimming away. Since their life cycles are relatively short, changes in their populations can be noted relatively quickly. A ratio where Group I is predominant would indicate a healthy environment. In contrast, a ratio where Group III is predominate would indicate an unhealthy or polluted environment. (Virginia.gov)
Flow Rate
The flow of a stream is directly related to the amount of water moving off the watershed into the stream channel. The flow rate is affected by weather, increasing during rainstorms and decreasing during dry periods. It also changes during different seasons of the year, decreasing during the summer months when evaporation rates are high and shoreline vegetation is actively growing and removing water from the ground. August and September are usually the months of lowest flow for most streams and rivers in most of the country. Flow is a function of water volume and velocity. It is important because of its impact on water quality and on the living organisms and habitats in the stream. Large, swiftly flowing rivers can receive pollution discharges and barely be affected, whereas small streams have less capacity to dilute and degrade wastes. Stream velocity, which increases as the volume of the water in the stream increases, determines the kinds of organisms that can live in the stream (some need fast-flowing areas; others need quiet pools). It also affects the amount of silt and sediment carried by the stream. Sediment introduced to quiet, slow-flowing streams will settle quickly to the stream bottom. Fast moving streams will keep sediment suspended longer in the water column. Lastly, fast-moving streams generally have higher levels of dissolved oxygen than slow streams because they are better aerated. (EPA)
Which parameters are more important in determining health.
DO
Usually streams with high dissolved oxygen concentrations greater than 7.5 mg/L are considered healthy streams. They are able to support a greater diversity of aquatic organisms. In general, DO levels less than 3 mg/L are stressful to most aquatic organisms.
pH
A pH higher or lower than the 6-8 range can decrease the survival rate of aquatic organisms.
Ammonia
Ammonia levels greater than approximately 0.1 mg/L usually indicate polluted waters. When levels reach 0.06 mg/L, fish can suffer gill damage.
Phosphates
Appropriate reference levels for total phosphate range from 0.01 to 0.075 mg/L, depending on the ecoregion. (U.S. EPA, 2002)
Nitrates
Appropriate reference levels range from 0.12 to 2.2 mg/L total. (U.S. EPA, 2002)
E.Coli
The Environmental Protection Agency had designated a federal standard that no single samples shall exceed 235 colonies/100 mL. If a sample exceeds the regulatory size, then the water is ruled as “contaminated"
The Virginia Department of Environmental Quality:
Parameter: Below Standard (4 mg/L)
Dissolved Oxygen 0.4% (+/- 0.4%)
Parameter: Below Standard (pH 6). Above Standard (pH 9).
pH 6.3% (+/- 3.1%) 0.0% (+/- 0.0%)
Parameter: Above Standard (31/32 degrees Celsius).
Temperature 0.0% (+/- 0.0%)
Parameter: Above Standard (235 CFU/100 mL)
E. coli 20.8% (+/- 6.6%)
Total Nitrogen (mg/L): Optimal <1, Suboptimal >2
Total Phosphorus (mg/L): Optimal <0.02, Suboptimal >0.05
-------------------------------------
Optimal environment for the JRSM
Habitat requirements: The James River Spinymussel lives in stream sites that vary in width from 10-75 feet and depth of 1/2 to 3 feet. It requires a slow to moderate water current with clean sand and cobble bottom sediments. The spinymussel is limited to areas of unpolluted water and may be more susceptible to competition from exotic clam species when its habitat is disturbed.